Some lifelong smokers do not develop lung cancer (Peto et al., 2000). The risk of developing lung cancer might vary as a function of smokers’ uptake and metabolism of carcinogens in tobacco smoke. NNK, a known carcinogen in rodents, is metabolically converted to NNAL, a human carcinogen (Hecht, 1998). Another common tobacco metabolite found in humans is cotinine; researchers often measure cotinine to determine exposure to tobacco smoke. This week’s ASHES reviews a study that examines the relationship between the urine levels of two carcinogen metabolites (NNAL and cotinine) and the risk for developing lung cancer (Yuan et al., 2009).
Methods:
- Participants were drawn from two cohorts:
1. Shanghai Cohort (Ross et al., 1992):
-
- At baseline, researchers assessed tobacco use and collected urine samples among 18,244 participants.
-
-
- 259 participants were diagnosed with lung cancer.
-
-
-
- Participants with lung cancer (cases) were matched to cancer free participants (controls) based on the following criteria: age within 2 years, date of urine collection within 1 month, and residential neighborhood at baseline.
-
2. Singapore Cohort (Hankin et al., 2001):
-
- At baseline, researchers assessed tobacco use among 63, 257 participants. The current research team requested urine samples from all surviving participants.
- 99 participants were diagnosed with lung cancer.
- Controls were matched to cases based on the following criteria: gender, dialect group, baseline age within 3 years, date of baseline interview within 2 years, and date of urine collection within 6 months.
- At baseline, researchers assessed tobacco use among 63, 257 participants. The current research team requested urine samples from all surviving participants.
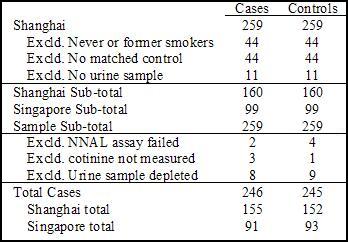
- Table 1 shows the exclusion of participants for the current study.
- Cases had a mean age of 65.6 (SD=6.8) years; controls matched at the time of cases’ cancer diagnosis had a mean age of 65.5 (SD=6.9) years.
- Urine samples were taken pre-lung cancer diagnosis, with an average of 4.0 (SD=2.5) years prior.
- The dependent measures were NNAL and cotinine levels.
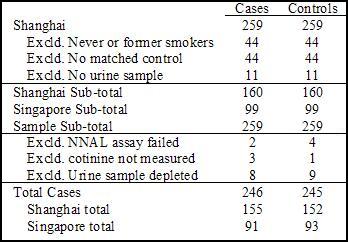
Figure 1. Study Sample Cases and Matched Controls. Click image to enlarge.
Results:
- Figure 1 shows the odds ratios of the joint effects of cotinine and NNAL levels on the risk of developing lung cancer.
- Compared to the low NNAL-low cotinine tertile, all odds ratios showed that the other tertiles were at a significantly increased risk for developing lung cancer.
- Participants in the highest tertile (high NNAL and cotinine levels) had an 8.47 fold (95% CI, 3.69-19.46) increased risk for developing lung cancer compared to participants in the lowest tertile (low NNAL and cotinine levels).
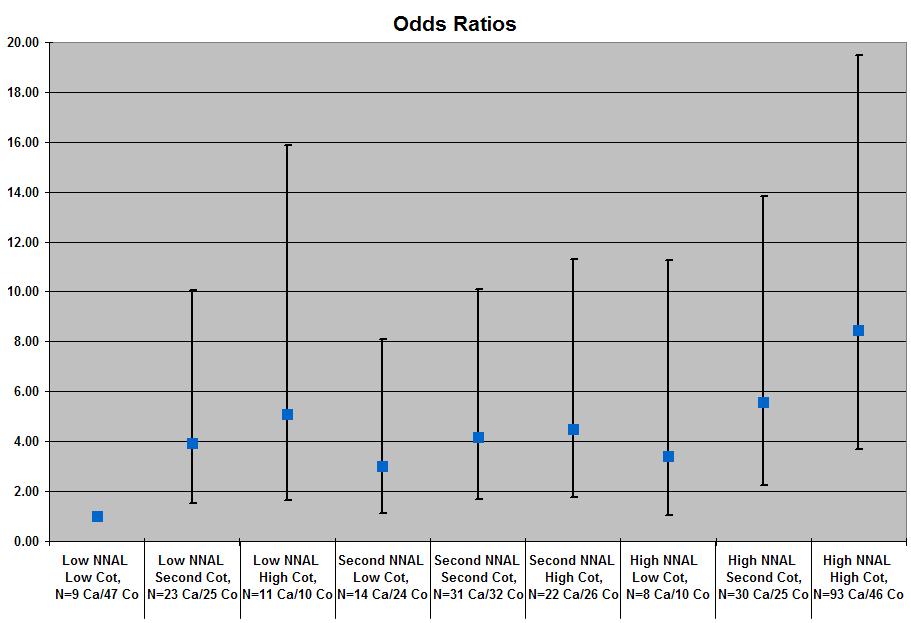
Figure 2. Odds Ratios of NNAL and Cotinine Levels Risk for Developing Lung Cancer (adapted from Yuan et al., 2009). Click image to enlarge.
Note. Cot stands for cotinine. Ca stands for cases. Co stands for controls.
The tertile cutoff values of NNAL for the Shanghai cohort were ≤0.105, 0.106 to 0.209, and ≥ 0.210 pmol/mg creatinine; for the Singapore cohort, ≤ 0.468, 0.469 to 0.819, and ≥ 0.820 pmol/mg creatinine. ORs were adjusted for age, year of interview, year of sample collection, gender and dialect group, cohort location, number of curettes smoked per day, and number of years smoking.
Limitations:
- This study uses self-report to determine smoking history.
- The concentration of NNK varies in cigarette brands (e.g., lower NNK concentrations in cigarettes from the Shanghai cohort), and brands were not controlled in this study.
- In the Shanghai sample, there was significantly less intense smoking behavior in the control group compared with the case group at baseline; consequently, the results might emerge from smoking intensity and not metabolic activity.
Discussion:
The results of this study show that the extensive animal data (Hecht, 1998) suggesting a link between NNK and cancer also might apply to humans. However, more research is needed to confirm the relationship between the metabolites reviewed above and the risk for developing lung cancer. This study suggests that measuring metabolite levels might offer a more accurate way to assess the risk of developing lung cancer for smokers than an assessment based on self-reported smoking history.
-Tasha Chandler
References:
Hankin, J. H., Stram, D. O., Arakawa, K., Park, S., Low, S. H., Lee, H. P., et al. (2001). Singapore Chinese Health Study: Development, validation, and calibration of the quantitative food frequency questionnaire. Nutrition and Cancer, 39(2), 187-195.
Hecht, S. S. (1998). Biochemistry, biology, and carcinogenicity of tobacco-specific N-nitrosamines. Chemical Research in Toxicology, 11(6), 559-603.
Peto, R., Darby, S., Deo, H., Silcocks, P., Whitley, E., & Doll, R. (2000). Smoking, smoking cessation, and lung cancer in the UK since 1950: Combination of national statistics with two case-control studies. BMJ (British Medical Journal), 321(7257), 323-329.
Ross, R. K., Yuan, J. M., Yu, M. C., Wogan, G. N., Qian, G. S., Tu, J. T., et al. (1992). Urinary aflatoxin biomarkers and risk of hepatocellular carcinoma. Lancet, 339(8799), 943-946.
Yuan, J. M., Koh, W. P., Murphy, S. E., Fan, Y., Wang, R., Carmella, S. G., et al. (2009). Urinary levels of tobacco-specific nitrosamine metabolites in relation to lung cancer development in two prospective cohorts of cigarette smokers. Cancer Research, 69(7), 2990-2995.
What do you think? Please use the comment link below to provide feedback on this article.




Sue May 6, 2009
This has to be the best BASIS title ever. Whoever thought that up is a genius. Keep up the good work.
Sue