There is reason to believe that feelings of excitement while gambling reinforce maladaptive gambling behavior among a subtype of pathological gamblers (PGs). These individuals crave excitement to a greater extent than other PGs and recreational gamblers and, for them, gambling might be especially rewarding because it offers excitement (Vachon & Bagby, 2009). Though the neurotransmitter dopamine is linked to behavior reinforcement and feelings of “sustained interest and approach” (Marco, 2010), the role of dopamine and excitement among pathological gamblers is unclear. This week, the WAGER reviews a study that examined the association between dopamine release and feelings of excitement during gambling to explore the potentially reinforcing role of dopamine in pathological gamblers’ maladaptive behavior in comparison to healthy controls (Linnet, Møller, Peterson, Gjedde, & Doudet, 2011).
Method
- Researchers recruited 18 Pathological Gamblers (PG) and 16 age-matched, non-gambling Healthy Controls (HC) (100% male; M age = 33.6, SD = 9.3; M age = 31.7, SD = 8.0, respectively). The researchers recruited the PG group from the Center of Pathological Gambling in Odense, Denmark based on referrals of those who were still considered to be active and pathological gamblers. They recruited the HC group through local newspaper ads.
- Researchers screened all participants before the study using the DSM-IV for Axis I psychopathology; this screen included a section for pathological gambling. They excluded any participants with present psychopathology (including substance abuse disorders) or neurological disorders that disqualified them from brain scanning procedures. Any HC participants who met the criteria for more than one symptom on the pathological gambling section of the DSM-IV were excluded.
- All research participants completed the following assessments:
- The Iowa Gambling Task (IGT) – a computerized card game consisting of four decks: two “disadvantageous” (i.e., selecting a card from these decks results in high initial gain but also high punishment and overall net loss) and two “advantageous” (i.e., smaller increments of gain but results in overall net gain). Participants completed three versions of the IGT; it became increasingly difficult for participants to distinguish between the advantageous and disadvantageous decks. Investigators measured performance as the number of cards selected from advantageous decks minus the number of cards selected from disadvantageous decks (calculated across five blocks of 20 trials for each of the three IGT versions, for a total of 300 cards).
- Self-reported excitement level– Participants rated their excitement levels after each IGT version by responding to the question “How exciting do you think this game is right now?” on a visual scale ranging from 1 (lowest) to 10 (highest).
- PET/MRI Scans– Participants underwent two 60-minute PET/MRI scans: one during the baseline condition, in which a computer explicitly told them which cards to choose, and another during the gambling condition, in which they chose cards for themselves. The researchers measured baseline dopamine binding potentials (BPND) and changes in binding potentials (ΔBPND) between the baseline and gambling scans. Lower binding potentials signify an increase in dopamine release during gambling).
- The researchers were primarily concerned with self-reported excitement levels as a function of group status (i.e., PG versus HC), condition (i.e., baseline versus gambling) and whether participants experienced increased dopamine release during the gambling tasks.
Results
- The researchers classified all participants according to whether they experienced increased dopamine release during the gambling tasks, resulting in four groups: PG with dopamine release (n= 8), HC with dopamine release (n=7), PG with dopamine inhibition (n=10), and HC with dopamine inhibition (n=9).
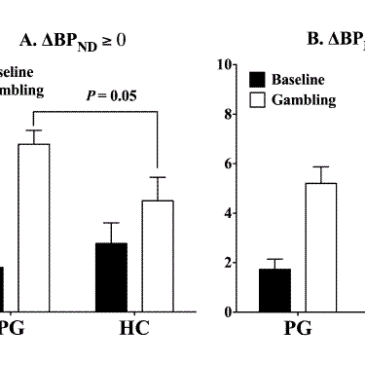
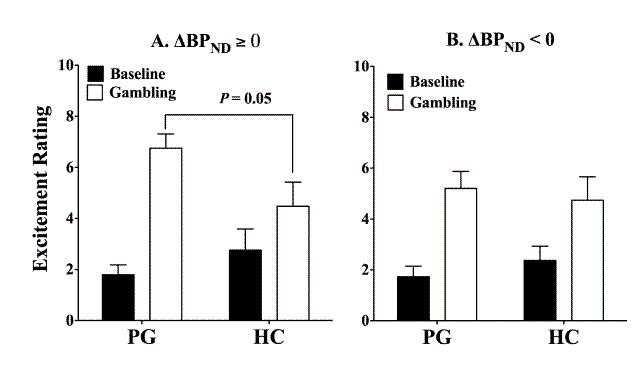
- As Figure 1 shows, the PGs with higher dopamine release during the gambling task reported a greater increase in excitement from baseline to gambling than the HC who also evidenced dopamine release t (13) = 3.28, P < 0.01. These PG also reported more excitement during the gambling task, t (13) = 2.24, P = 0.05. There was no difference at baseline.
Figure 1- Excitement levels in PG and HC during the baseline and gambling conditions. Notes: “Δ BP ND ≥ 0” indicates increased dopamine release during gambling. “Δ BP ND < 0” indicates dopamine inhibition during gambling. (Adapted from Linnet, Møller, Peterson, Gjedde, & Doudet, 2011).
- Two-way ANOVAS revealed that PG with more dopamine release had greater excitement levels across all three IGT games, F (2, 12) = 10.69, P < .005.
- Among all PGs, the change in dopamine release from non-gambling to gambling were significantly correlated with changes in excitement level, t (18) = 0.52, P < 0.05. This relationship was not evident among HCs.
- There were no significant correlations between PG or HC’s increased excitement level and performance on the IGT, or between change in dopamine release and their IGT performance, but PG had lower performance than HC overall.
Limitations
- This study’s all-male, small sample as well as the screening of participants for co-occurring substance abuse disorders limits the generalizability of these findings. A large proportion of PGs in the community also experience co-occurring substance and other disorders.
- This study also restricted its exploration of dopamine transmission to the ventral striatum brain region, which is only one of the possible areas related to dopamine release in pathological gamblers.
- Finally, because dopamine serves other functions besides reinforcing behavior, such as warning for potentially undesirable events, this study does not conclusively prove that changes in dopamine release indicate behavior reinforcement. Instead, PGs might experience higher dopamine release because they are expecting a potentially negative outcome from gambling.
Discussion
This study found an association between dopamine release in the ventral striatum and increased excitement levels during a gambling task among some pathological gamblers. The authors suggest that dopamine release causes a “double deficit” for these PGs, such that dopamine both reinforces gambling (e.g., by increasing feelings of excitement) and promotes risky decision making. The authors speculate about the role of dopamine release in chemical forms of addiction, where people also perform maladaptive behaviors (e.g., repeated drug use) despite negative consequences. In some chemical forms of addiction, however, individuals often show a “blunted” dopamine response (e.g., Volkow, Wang, Fowler, & Logan, 1997). With regard to dopamine, similarities and differences with chemical addiction warrant further research. Moreover, dopamine might not be the only factor involved in the excitement levels among pathological gamblers, and future studies should explore the reinforcing role of other biological factors.
-Brittany Bannon
What do you think? Please use the comment link below to provide feedback on this article.
References
Linnet, J., Møller, A., Peterson, E., Gjedde, A., & Doudet, D. (2011). Dopamine release in ventral striatum during Iowa Gambling Task performance is associated with increased excitement levels in pathological gambling. Addiction, 106(2), 383-390.
Marco, L., (2010). The neurobiology of desire: Dopamine and the regulation of mood and motivational states in humans. In Kringelbach, M. L., & Berridge, K. C. (Eds.) Pleasures of the Brain (pp. 222-243). New York, NY: Oxford University Press.
Moodie, C., & Finnigan, F. (2005). A comparison of the autonomic arousal of frequent,
infrequent and non-gamblers while playing fruit machines. Addiction, 100, 51-59.
Vachon, D. D., & Bagby, R. M. (2009). Pathological gambling subtypes. Psychological Assessment, 21(4), 608-615.
Volkow, N. D., Wang, G. J., Fowler, J. S., & Logan, J. (1997). Decreased striatal dopaminergic responsiveness in detoxified cocaine-dependent subjects. Nature, 386(6627), 830-833.